For years, a significant number of racers have been running alternative fuels. Historically, the most popular has been “alcohol,” which in the automotive context typically refers to methanol. Methanol has been used by USAC, IRL, Champ car, and NHRA Top Alcohol class racers through the years. It burns with a clear flame with minimal soot production and is very corrosive in its liquid form. Methanol is available from several sources such as Sunoco Racing Fuels, VP Racing Fuels, and Torco. Methanol is a relatively simple molecule that Starts Out looking like methane (CH4), but replaces one of the hydrogen legs with an —OH group that now qualifies it as an actual alcohol to chemists. Because the fuel carries some amount of oxy- gen within it, the stoichiometric bal- ance for clean combustion in air is shifted. If methanol were to be burned at the same mass ratio of 14.64:1 as most gasoline blends, there would just be too many oxygen molecules available for each hydro- gen and carbon in the mix. The result would be a lean burn with less- than-optimal power. To fix this more fuel is added per unit of air in Order to achieve a balance Of all the hydrogens, carbons, and oxygens (which may come from either the air or the fuel itself) during combustion. With methanol, this balance is a ratio of 6:1 in Earth’s atmosphere.
Methanol contains slightly less energy per unit, but because the stoi- chiometric ratio is so rich (relatively speaking, Of course!), more total fuel is burned on each cycle with the same amount Of airflow. Once the correct ratio of fuel is delivered, the loss of specific energy per unit volume becomes irrelevant, as more total energy can be released with methanol than with gasoline for the same airmass. The limiting factor on how much power most engines can deliver is usually their maximum air- flow limit. A limit on maximum airflow with a given target air/fuel ratio means a limit on total fuel deliv- ery without going excessively rich and hurting power. If there is a lim- ited amount of chemical energy to be released from the fuel, this also limits the potential mechanical energy out of the engine. With alcohol fuels that run at much richer mixtures in order to achieve the same chemical balance between the hydrogens, carbons, and oxygens, more total fuel can be burned with the same amount of out- side air being pumped through the engine. As long as the fuel system is able to keep up with the higher flow rate demands of an alcohol-based sys- tem, more total power can be deliv- ered even though the specific energy of the fuel may be slightly lower.
Alcohol fuels also tend to burn at a noticeably lower temperature than gasoline. This can greatly reduce loads on the cooling system thermal concerns within the exhaust system at high power. Making large amounts of power from an engine with a fixed pumping capability gets easier when the fuel itself is helping to deliver oxygen to the cylinders. A six-to-one ratio of air and fuel looks pretty rich at first glance. Keep in mind that this is actually the balance point. If burned at that ratio, the chemistry does not see it as “richer” than gasoline that is being burned at 14.64:1. Rather, it’s just a smaller mol- ecule that happens to be contributing to the oxygen source of the air during the reaction. A ratio of 9:1 may be extremely rich with gasoline, but with methanol it’s exceptionally lean. To make things more clear, let’s go back to using units of lambda. Lambda is just the excess air ratio for combustion. It does not care what type of fuel is being burned because it is really a reference of where things are at the moment compared to whatever the chemical balance point is. Equation 3.1 gave us the definition of lambda:
,In the case of alcohol blends as fuels, the stoichiometric air/fuel ratio is just different than what we’re used to seeing for gasoline. Using methanol’s stoichiometric balance point of 6:1, lambda can be calculated in much the same manner as with gasoline. If the actual ratio of 9:1 from our earlier example is to be observed in units of lambda, we find that the current excess air ratio is actually:
The resulting lambda of 1.50 is so lean that combustion may not occur at all due to being near the lean igni- tion limit of the fuel, depending upon conditions. To get a clean burn, much more fuel is required. In this case, 50% more fuel would be neces- sary just to get back to the chemically ideal balance that is desirable at idle and cruise in most engines.
Oxygen Sensors and Alcohol Lambda sensors, both wideband or narrowband, do not know what
type of fuel is being burned in the cylinders. Frankly, they don’t care and neither should you. What they tell you is which side of the chemical balance the engine is on at any given point. With a wideband, you just have the added benefit of knowing how far on either side of the balance the engine is. Although this may sound like a bit of a handicap at first, it’s really a blessing in disguise.
Remember that the oxygen sen- sor is really only measuring oxygen content and not fuel content in the exhaust gases. The display that is returned for “air/fuel ratio” on many cheaper units is really just lambda multiplied by about 14.64 to give the less-educated user something he is used to seeing. If methanol is being burned at a ratio of 6.00:1 in an engine, the wideband sensor will detect a ratio of lambda=1.00. The cheaper units will display this as (1.00) x (14.64) for an indicated ratio
of 14.64:1. Obviously, there is a mis- match here. The problem arises from the assumption that the engine is burning gasoline when, in fact, it’s really burning methanol. The wide- band sensor is actually doing its job perfectly, it’s just that the display configuration is misleading. The best course of action is to configure the display to only show lambda. That way, the calibrator is not confused by the conversion factor. Most experi- enced calibrators are used to working in units of lambda anyway, so there is no problem with stopping here. If you really want to see an actual air/fuel ratio with methanol, the wideband output configuration must be modified to show lambda multiplied by the stoichiometric balance point of 6.00:1. Once you can get used to looking at units of lambda rather than air/fuel ratio, it becomes far easier to tune almost any alterna- tive fuel blend.
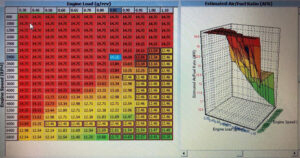
Calibration Setup for Alcohol So how does one set an EFI system up to properly control a methanol fueled engine? The answer is surpris- ingly simple. All that’s really needed is to open the target air/fuel ratio table and input new targets based upon the chemistry of the fuel being used. With methanol, most of Zones 1, 2, 3, 4, 5, and 7 are set to lambda—I .00, which is a ratio of in this case. When tuning the VE table, just look at the wideband output in units of lambda, not air/fuel ratio. Lambda will show the exact error if the target is stoichio- metric operation, so corrections to the VE surface can be made based upon lambda alone. Make corrections to the VE table in the same exact manner as with gasoline by using the delivered lambda as a correction factor.
When the time comes for WOT tuning with alcohol fuels, just keep working in units of lambda. The same lambda setpoints can be used with alcohol fuels as with gasoline. A naturally aspirated engine can be run at lambda—O.87. With alcohol just like gasoline, the only difference will be the actual commanded air/ fuel ratio. If the base fuel map does not support units of lambda, simply cal- culate the desired ratio using a vari- ant of Equation 3.1 as before: The target air/fuel ratio within Zone 6 can be populated with a ratio of 5.16:1 for use with methanol. This provides a safe starting point from which the WOT volumetric efficien- Cies can be calculated. Because alco- hol fuels burn cooler, it may be found that this much extra enrich- ment is not necessary. Running slightly leaner, closer to LBT, around lambda-0.92 may be possible with- out damage on alcohol-fueled engines. Only actual testing of the engine in question with a loaded dyno or at the track will tell if this approach is acceptable.
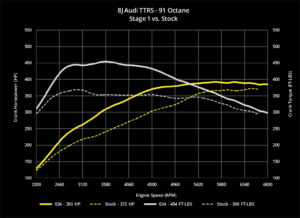
Alcohol-based fuels also have inherently high octane ratings. This high octane rating means that the fuel is not very likely to experience knock under higher loads and tem- peratures. Many alcohol engines will be able to run ignition angles all the way up to MBT timing at WOT with- out knock. Keep this in mind as test- ing is performed. Air/fuel ratios should still be corrected before opti- mizing ignition angle. Once the delivered air/fuel ratio matches the target, timing sweeps can be done to find the best power and torque for the engine. Don’t wait for spark knock to tell you when too much timing has been delivered, as this may be well past MBT with alcohol. Just watch the torque output as tim- ing is added. If delivered torque drops with added timing, chances are that MBT timing has been passed. Backing off the timing may actually deliver more torque output. Even in boost, timing may still be able to reach MBT with alcohol with- out knock. Don’t be afraid of what looks like large timing numbers com- pared to gasoline, but be careful not to go too far and lose power. Always try to run the minimum amount of ignition lead necessary to deliver maximum torque without knock or abnormal combustion contributes to the warming of the Earth, so many efforts have been made to reduce the overall genera- tion of it. By blending conventional gasoline with a percentage of ethanol, the carbon content of the fuel is reduced, which in turn reduces production of C02 by the vehicles burning this fuel. Much like methanol, ethanol starts out as a simple hydrocarbon molecule. In this case, the base mole- cule would be ethane (C2H6), which is a two-carbon chain. To make ethane into its alcohol equivalent, ethanol, an —OH group again replaces one of the hydrogen atoms on the molecule. The result is another oxy- gen-bearing fuel that has a different stoichiometric combustion balance point that is somewhere between that of gasoline and methanol. The Ethanol One of the unintended benefits of the environmental movement has been the availability of cheap race fuel. E85 is billed as a “greener” alternative to gasoline because its combustion results in lower C02 production. Carbon dioxide is a greenhouse gas that stoichiometric balance for clean com- bustion of pure ethanol is a mass ratio of 9.00:1. Ethanol itself has an inherently high octane rating due to the compact shape of the molecule. The octane rating of pure ethanol is approximately 129 RON (Research Octane Number, the higher of the two [MON, Motoring Octane Num- ber is the other]). So in a rare coinci- dence, the environmental movement has lead to widespread availability of cheap, high octane fuel. Many different blends of ethanol are available today. Pure ethanol is actually rare, and usually only avail- able as a research fuel with corre- spondingly high pricing. However, E85 has become far more common at gas stations today. E85 is typically a blend of 85% ethanol and 15% gaso- line. Adding the 15% gasoline to the mixture greatly helps cold tempera- ture evaporation rates and makes storage less of a concern for water absorption. The blend between ethanol and gasoline in E85 results in a stoichiometric balance point of a 9.85:1 mass ratio. When tuning an E85 application, simply use 9.85
instead of the 14.64 used for gasoline or 6.00 used for methanol for zones 1, 2, 3, 4, 5, and 7. For a naturally aspirated engine looking to run at lambda=0.86 at WOT, the actual tar- get ratio is found as before by:
Remember that even though this looks like an extremely rich ratio at first glance, it’s going to behave sim- ilar to a gasoline engine operating at about 12.6:1. Also remember that a cheaper wideband may actually dis- play “12.6” at this ratio when it really is measuring lambda=0.86 and converting it to a gasoline-based display number. Just stick with the lambda reading to avoid confusion during testing. E85 has an octane rat- ing of approximately 105 (R+M)/2, depending on the blend. This high octane rating is what makes this “green” fuel attractive for many rac- ing applications. In winter months, the fuel sold as “E85” is usually actually closer to E70, a 70% blend of ethanol that contains more gasoline. This is done to aid in cold starting of the engine since the alcohol fuels have lower vapor pres- sures than their gasoline equivalent and have trouble evaporating com- pletely at low temperatures. During the transition months, the fuel in the tanks at gas stations selling E85 may vary anywhere between these two blends, so it’s important to know what you’re burning before getting too far into the calibration exercises. Most OEM vehicles that are des- ignated as “flex fuel” vehicles have the ability to detect the difference between the seasonal blends of ethanol-based fuels and normal gaso- line. This is done either by a specific sensor or by a more complex logic within the ECU. The logic-based sys- tems employ a “virtual sensor” that solves for alcohol content by examin- ing fuel trims and knock sensor feed- back immediately after startup if the fuel level has changed. The logic used here is pretty involved and closely guarded by the manufacturers who have invented it, so it should not be expected to show up in the garden- variety standalone controller any time soon. The fuel composition sen- sor is typically installed either in the fuel tank or on the fuel feed line lead- ing to the engine and is active any- time the ECU is running. One such example is GM’s flex fuel sensor used in some truck applications. This sen- sor returns a P WM signal to the ECU that indicates both alcohol concen- tration percentage and fuel tempera- ture. If the ECU has the software to adjust fueling based on this input, it can make tuning with various blends of alcohol fuels a breeze.
Two primary hardware concerns must be addressed when running alcohol fuels: capacity and corrosion. Because these fuels require larger vol- umes of flow to reach their stoichio- metric and power enrichment ratios, the fuel system must be sized to sup- port these higher flow rates. It takes about 5()% more fuel volume to sup- port the same air/fuel ratio with E85 as it would gasoline. This means that the fuel pump, lines, filters, rails, and injectors all need to be sized accord- ingly. Before simply buying larger capacity components, it’s also impor- tant to consider the materials used. Alcohol fuels can be corrosive to the parts in many older fuel systems. Rubber and EPDM seals can literally be dissolved by alcohols, and the deposits may clog filters and injec- tors. Viton, Teflon, or another equiv- alent material should be used instead. Aluminum will oxidize in the presence of alcohols too, so stainless steel fittings, rails, and hard lines should be used whenever possi- ble with alcohol fuels.
Pump Gas Even those of you running stan- dard pump gasoline are probably get- ting a mild alcohol blend. By law, most gas stations today are now allowed to sell blends of as much as 1()% ethanol as “gasoline.” The ethanol is being used to displace the MTBE (methyl tert-butyl ether), which has its own environmental and health concerns. It used to be that stations were required to post a sticker on the pump declaring the alcohol content, but that’s no longer happening. Some regions were quicker to adopt the maximum allowable ethanol content in the pump gasoline. In my travels and discussions with shop owners and calibrators across the country, I’ve found that almost every station can be assumed to be selling some alco- hol blend unless stated otherwise. In most cases, this means assuming a blend of 10% ethanol. Any addition of alcohol blends to the gasoline will shift the stoi- chiometric balance point. The more oxygen carried by the fuel, the richer the stoichiometric point will be.Where E85 has a stoichiometric ratio of 9.85:1, EIO (most pump gas blends) will balance at about 14.2:1. This means that tuning to an assumed pure gasoline composition with a stoichiometric point of 14.64:1 may include a 3% error before the engine is ever started. The wideband oxygen sensor will still typically display at lambda—I .00, even though this is really an actual ratio of 14.2:1 with a 10% ethanol blend. Three percent may not sound like much, and it really isn’t, but the idea behind engine calibration is to get all vari- ables as close to optimal as possible. Leaving a 3% error in the fuel’s stoi- chiometric point just makes tuning the volumetric efficiency table, startup fuel, and transient fueling that much less accurate. It’s a good idea to just adjust at the beginning before baking that error into every other calculation later on. The only trick with EIO gasoline blends is that they must be treated like normal gasoline for the purposes of spark tuning. The alcohol in the fuel is part of the cocktail of addi- tives used to get the desired octane rating. In this case, 91 octane really is just 91 octane, so the same care must be taken to avoid knock at high loads with EIO blends as is typically done with conventional gasoline.